 Subthreshold membrane phenomena
Subthreshold membrane phenomena



 Subthreshold membrane phenomena
Subthreshold membrane phenomena
3.1 INTRODUCTION
In the previous chapter the subthreshold behavior of the nerve cell was discussed qualitatively. This chapter describes the physiological basis of the resting voltage and the subthreshold response of an axon to electric stimuli from a quantitative perspective.
where the subscript k refers to an arbitrary kth ion. Transposing terms in Equation 3.15 gives
Since the membrane is extremely thin, we can consider any small patch as planar and describe variations across it as one-dimensional (along a normal to the membrane). If we call this direction x, we may write out Equation 3.16 as
Equation 3.17 can be rearranged to give
Equation 3.18 may now be integrated from the intracellular space (i) to the extracellular space (o); that is:
Carrying out the integrations in Equation 3.19 gives
where ci,k and co,k denote the intracellular and extracellular concentrations of the kth ion, respectively. The equilibrium voltage across the membrane for the kth ion is, by convention, the intracellular minus the extracellular potential (Vk = Fi - Fo), hence:
Equation 3.21 is the famous Nernst equation derived by Walther Hermann Nernst in 1888 (Nernst, 1888). By Substituting 37 °C which gives T = 273 + 37 and +1 for the valence, and by replacing the natural logarithm (the Napier logarithm) with the decadic logarithm (the Briggs logarithm), one may write the Nernst equation for a monovalent cation as:
At room temperature (20 °C), the coefficient in Equation 3.22 has the value of 58; at the temperature of seawater (6 °C), it is 55. The latter is important when considering the squid axon.
Example
We discuss the subject of equilibrium further by means of the example described in Figure 3.2, depicting an axon lying in a cylindrical experimental chamber. The potential inside the axon may be changed with three interchangeable batteries (A, B, and C) which may be placed between the intracellular and extracellular spaces. We assume that the intracellular and the extracellular spaces can be considered isopotential so that the transmembrane voltage Vm (difference of potential across the membrane) is the same everywhere. (This technique is called voltage clamp, and explained in more detail in Section 4.2.) Furthermore, the membrane is assumed to be permeable only to potassium ions. The intracellular and extracellular concentrations of potassium are ci,K and co,K, respectively. In the resting state, the membrane voltage Vm (= Fi - Fo) equals VK, the Nernst voltage for K+ ions according to Equation 3.21.
 The membrane plays an important role in establishing the resting and active electric properties of an excitable cell, through its regulation of the movement of ions between the extracellular and intracellular spaces. The word ion (Greek for "that which goes") was introduced by Faraday (1834). The ease with which an ion crosses the membrane, namely the membrane permeability, differs among ion species; this selective permeability will be seen to have important physiological consequences. Activation of a cell affects its behavior by altering these permeabilities. Another important consideration for transmembrane ion movement is the fact that the ionic composition inside the cell differs greatly from that outside the cell. Consequently, concentration gradients exist for all permeable ions that contribute to the net ion movement or flux. The principle whereby ions flow from regions of high to low concentration is called diffusion.
The membrane plays an important role in establishing the resting and active electric properties of an excitable cell, through its regulation of the movement of ions between the extracellular and intracellular spaces. The word ion (Greek for "that which goes") was introduced by Faraday (1834). The ease with which an ion crosses the membrane, namely the membrane permeability, differs among ion species; this selective permeability will be seen to have important physiological consequences. Activation of a cell affects its behavior by altering these permeabilities. Another important consideration for transmembrane ion movement is the fact that the ionic composition inside the cell differs greatly from that outside the cell. Consequently, concentration gradients exist for all permeable ions that contribute to the net ion movement or flux. The principle whereby ions flow from regions of high to low concentration is called diffusion.
 One consequence of this ion flow is the tendency for ions to accumulate at the inner and outer membrane surfaces, a process by which an electric field is established within the membrane. This field exerts forces on the ions crossing the membrane since the latter carry an electric charge. Thus to describe membrane ion movements, electric-field forces as well as diffusional forces should be considered. Equilibrium is attained when the diffusional force balances the electric field force for all permeable ions.
One consequence of this ion flow is the tendency for ions to accumulate at the inner and outer membrane surfaces, a process by which an electric field is established within the membrane. This field exerts forces on the ions crossing the membrane since the latter carry an electric charge. Thus to describe membrane ion movements, electric-field forces as well as diffusional forces should be considered. Equilibrium is attained when the diffusional force balances the electric field force for all permeable ions.
 For a membrane that is permeable to only one type of ion, equilibrium requires that the force due to the electric field be equal and opposite to the force due to diffusion. In the next section we shall explore the Nernst equation, which expresses the equilibrium voltage associated with a given concentration ratio. Equilibrium can also be defined by equating the electrochemical potential on both sides of the membrane.
For a membrane that is permeable to only one type of ion, equilibrium requires that the force due to the electric field be equal and opposite to the force due to diffusion. In the next section we shall explore the Nernst equation, which expresses the equilibrium voltage associated with a given concentration ratio. Equilibrium can also be defined by equating the electrochemical potential on both sides of the membrane.
 The Nernst equation is derived from two basic concepts involving ionic flow - those resulting from an electric field force and those resulting from a diffusional force. A more rigorous thermodynamic treatment is available, and the interested reader should consult references such as van Rysselberghe (1963) and Katchalsky and Curran (1965).
The Nernst equation is derived from two basic concepts involving ionic flow - those resulting from an electric field force and those resulting from a diffusional force. A more rigorous thermodynamic treatment is available, and the interested reader should consult references such as van Rysselberghe (1963) and Katchalsky and Curran (1965).
 We shall also derive the Goldman-Hodgkin-Katz equation, which gives the steady-state value of the membrane voltage when there are several types of ions in the intracellular and extracellular media, and when the membrane is permeable to all of them. As will be seen, the Goldman-Hodgkin-Katz equation is a straightforward extension of the Nernst equation.
We shall also derive the Goldman-Hodgkin-Katz equation, which gives the steady-state value of the membrane voltage when there are several types of ions in the intracellular and extracellular media, and when the membrane is permeable to all of them. As will be seen, the Goldman-Hodgkin-Katz equation is a straightforward extension of the Nernst equation.
 A more detailed discussion of physical chemistry, which contributes to many topics in this chapter, can be found in standard textbooks such as Edsall and Wyman (1958) and Moore (1972).
A more detailed discussion of physical chemistry, which contributes to many topics in this chapter, can be found in standard textbooks such as Edsall and Wyman (1958) and Moore (1972).


(3.15)


(3.16)


(3.17)


(3.18)


(3.19)


(3.20)


(3.21)
where Vk = equilibrium voltage for the kth ion across the membrane Fi - Fo i.e., the Nernst voltage [V] R = gas constant [8.314 J/(mol·K)] T = absolute temperature [K] zk = valence of the kth ion F = Faraday's constant [9.649 × 104 C/mol] ci,k = intracellular concentration of the kth ion co,k = extracellular concentration of the kth ion


(3.22)
 In Figure 3.2 the vertical axis indicates the potential F, and the horizontal axis the radial distance r measured from the center of the axon. The membrane is located between the radial distance values ri and ro. The length of the arrows indicates the magnitude of the voltage (inside potential minus outside potential). Their direction indicates the polarity so that upward arrows represent negative, and downward arrows positive voltages (because all the potential differences in this example are measured from negative potentials). Therefore, when DV is positive (downward), the transmembrane current (for a positive ion) is also positive (i.e., outward).
In Figure 3.2 the vertical axis indicates the potential F, and the horizontal axis the radial distance r measured from the center of the axon. The membrane is located between the radial distance values ri and ro. The length of the arrows indicates the magnitude of the voltage (inside potential minus outside potential). Their direction indicates the polarity so that upward arrows represent negative, and downward arrows positive voltages (because all the potential differences in this example are measured from negative potentials). Therefore, when DV is positive (downward), the transmembrane current (for a positive ion) is also positive (i.e., outward).
 A. Suppose that the electromotive force emf of the battery A equals VK. In this case Vm = VK and the condition corresponds precisely to the one where equilibrium between diffusion and electric field forces is achieved. Under this condition no net flow of potassium ions exists through the membrane (see Figure 3.2A). (The flow through the membrane consists only of diffusional flow in both directions.)
A. Suppose that the electromotive force emf of the battery A equals VK. In this case Vm = VK and the condition corresponds precisely to the one where equilibrium between diffusion and electric field forces is achieved. Under this condition no net flow of potassium ions exists through the membrane (see Figure 3.2A). (The flow through the membrane consists only of diffusional flow in both directions.)
 B. Suppose, now, that the voltage of battery B is smaller than VK (|Vm < VK|). Then the potential inside the membrane becomes less negative, a condition known as depolarization of the membrane. Now the electric field is no longer adequate to equilibrate the diffusional forces. This imbalance is DV = Vm - VK and an outflow of potassium (from a higher electrochemical potential to a lower one) results. This condition is illustrated in Figure 3.2B.
B. Suppose, now, that the voltage of battery B is smaller than VK (|Vm < VK|). Then the potential inside the membrane becomes less negative, a condition known as depolarization of the membrane. Now the electric field is no longer adequate to equilibrate the diffusional forces. This imbalance is DV = Vm - VK and an outflow of potassium (from a higher electrochemical potential to a lower one) results. This condition is illustrated in Figure 3.2B.
 C. If, on the other hand, battery C is selected so that the potential inside the membrane becomes more negative than in the resting state (|Vm| > |VK|), then the membrane is said to be hyperpolarized. In this case ions will flow inward (again from the higher electrochemical potential to the lower one). This condition is described in Figure 3.2C.
C. If, on the other hand, battery C is selected so that the potential inside the membrane becomes more negative than in the resting state (|Vm| > |VK|), then the membrane is said to be hyperpolarized. In this case ions will flow inward (again from the higher electrochemical potential to the lower one). This condition is described in Figure 3.2C.

Fig. 3.2. An example illustrating the Nernst equation and ion flow through the membrane in
 (A) equilibrium at rest,
(A) equilibrium at rest,
 (B) depolarized membrane, and
(B) depolarized membrane, and
 (C) hyperpolarized membrane.
(C) hyperpolarized membrane.
 The diffusional force arising from the concentration gradient is equal and opposite to the equilibrium electric field VK which, in turn, is calculated from the Nernst potential (see Equation 3.21). The Nernst electric field force VK is described by the open arrow. The thin arrow describes the actual electric field Vm across the membrane that is imposed when the battery performs a voltage clamp (see Section 4.2 for the description of voltage clamp). The bold arrow is the net electric field driving force DV in the membrane resulting from the difference between the actual electric field (thin arrow) and the equilibrium electric field (open arrow).
The diffusional force arising from the concentration gradient is equal and opposite to the equilibrium electric field VK which, in turn, is calculated from the Nernst potential (see Equation 3.21). The Nernst electric field force VK is described by the open arrow. The thin arrow describes the actual electric field Vm across the membrane that is imposed when the battery performs a voltage clamp (see Section 4.2 for the description of voltage clamp). The bold arrow is the net electric field driving force DV in the membrane resulting from the difference between the actual electric field (thin arrow) and the equilibrium electric field (open arrow).
  | (3.23) |
Note that Equation 3.23 reflects the fact that all ions are univalent and that chloride is negative. The condition represented by Equation 3.23 is that all ions are in equilibrium; it is referred to as the Donnan equilibrium.

Fig. 3.4. An electric circuit representation of a membrane patch. In this diagram, VNa, VK, and VL represent the absolute values of the respective emf's and the signs indicate their directions when the extracellular medium has a normal composition (high Na and Cl, and low K, concentrations).
3.4.2 The Value of the Resting Voltage, Goldman-Hodgkin-Katz Equation
The relationship between membrane voltage and ionic flux is of great importance. Research on this relationship makes several assumptions: first, that the biological membrane is homogeneous and neutral (like very thin glass); and second, that the intracellular and extracellular regions are completely uniform and unchanging. Such a model is described as an electrodiffusion model. Among these models is that by Goldman-Hodgkin-Katz which is described in this section.
This approximation was originally introduced by David Goldman (1943).
for the kth ion flux. If we now insert the constant field approximation of Equation 3.24 (dF/dx = Vm/h) the result is
(To differentiate ionic concentration within the membrane from that outside the membrane (i.e., inside versus outside the membrane), we use the symbol cm in the following where intramembrane concentrations are indicated.) Rearranging Equation 3.26 gives the following differential equation:
We now integrate Equation 3.27 within the membrane from the left-hand edge (x = 0) to the right-hand edge (x = h). We assume the existence of resting conditions; hence each ion flux must be in steady state and therefore uniform with respect to x. Furthermore, for Vm to remain constant, the total transmembrane electric current must be zero. From the first condition we require that jk(x) be a constant; hence on the left-hand side of Equation 3.27, only ckm(x) is a function of x. The result of the integration is then
Equation 3.28 can be solved for jk, giving
The concentrations of the kth ion in Equation 3.29 are those within the membrane. However, the known concentrations are those in the intracellular and extracellular (bulk) spaces. Now the concentration ratio from just outside to just inside the membrane is described by a partition coefficient, b. These are assumed to be the same at both the intracellular and extracellular interface. Consequently, since x = 0 is at the extracellular surface and x = h the intracellular interface, we have
then
In Equation 3.34 the expression for sodium ion current is seen to be similar to that for potassium (except for exchanging Na for K); however, the expression for chloride requires, in addition, a change in sign in the exponential term, a reflection of the negative valence.
Multiplying through by the permeabilities and collecting terms gives:
From this equation, it is possible to solve for the potential difference Vm across the membrane, as follows:
where Vm evaluates the intracellular minus extracellular potential (i.e., transmembrane voltage). This equation is called the Goldman-Hodgkin-Katz equation. Its derivation is based on the works of David Goldman (1943) and Hodgkin and Katz (1949). One notes in Equation 3.37 that the relative contribution of each ion species to the resting voltage is weighted by that ion's permeability. For the squid axon, we noted (Section 3.5.2) that PNa/PK = 0.04, which explains why its resting voltage is relatively close to VK and quite different from VNa.
Example
It is easy to demonstrate that the Goldman-Hodgkin-Katz equation (Equation 3.37) reduces to the Nernst equation (Equation 3.21). Suppose that the chloride concentration both inside and outside the membrane were zero (i.e., co,Cl = ci,Cl = 0). Then the third terms in the numerator and denominator of Equation 3.37 would be absent. Suppose further that the permeability to sodium (normally very small) could be taken to be exactly zero (i.e., PNa = 0). Under these conditions the Goldman-Hodgkin-Katz equation reduces to the form of the Nernst equation (note that the absolute value of the valence of the ions in question |z| = 1). This demonstrates again that the Nernst equation expresses the equilibrium potential difference across an ion permeable membrane for systems containing only a single permeable ion.
3.4.3 The Reversal Voltage
The membrane potential at which the (net) membrane current is zero is called the reversal voltage (VR). This designation derives from the fact that when the membrane voltage is increased or decreased, it is at this potential that the membrane current reverses its sign. When the membrane is permeable for two types of ions, A+ and B+, and the permeability ratio for these ions is PA/PB, the reversal voltage is defined by the equation:
This equation resembles the Nernst equation (Equation 3.21), but it includes two types of ions. It is the simplest form of the Goldman-Hodgkin-Katz equation (Equation 3.37).
based on the definition of V' given above.
3.6.2 The Steady-State Response
We first consider the stationary case (i.e., d/dt = 0) which is the steady-state condition achieved following the application of current step. This corresponds to the limit t
From the current conservation laws, it follows also that the transmembrane current per unit length, im, must be related to the loss of Ii or to the gain of Io as follows:
Note that this expression is consistent with Ii + Io = 0. The selection of the signs in Equation 3.42 is based on outward-flowing current being defined as positive. From these definitions and Equations 3.40 and 3.41 (and recalling that V' = Fi - Fo - Vr), it follows that
Furthermore, by differentiating with respect to x, we obtain:
 In view of the very small thickness of a biological membrane as compared to its lateral extent, we may treat any element of membrane under consideration as planar. The Goldman-Hodgkin-Katz model assumes, in fact, that the membrane is uniform, planar, and infinite in its lateral extent. If the x-axis is chosen normal to the membrane with its origin at the interface of the membrane with the extracellular region, and if the membrane thickness is h, then x = h defines the interface of the membrane with the intracellular space. Because of the assumed lateral uniformity, variations of the potential field F and ionic concentration c within the membrane are functions of x only. The basic assumption underlying the Goldman-Hodgkin-Katz model is that the field within the membrane is constant; hence
In view of the very small thickness of a biological membrane as compared to its lateral extent, we may treat any element of membrane under consideration as planar. The Goldman-Hodgkin-Katz model assumes, in fact, that the membrane is uniform, planar, and infinite in its lateral extent. If the x-axis is chosen normal to the membrane with its origin at the interface of the membrane with the extracellular region, and if the membrane thickness is h, then x = h defines the interface of the membrane with the intracellular space. Because of the assumed lateral uniformity, variations of the potential field F and ionic concentration c within the membrane are functions of x only. The basic assumption underlying the Goldman-Hodgkin-Katz model is that the field within the membrane is constant; hence


(3.24)
where F0 = potential at the outer membrane surface Fh = potential at the inner membrane surface Vm = transmembrane voltage h = membrane thickness  The Nernst equation evaluates the equilibrium value of the membrane voltage when the membrane is permeable to only one kind of ion or when all permeable ions have reached a Donnan equilibrium. Under physiological conditions, such an equilibrium is not achieved as can be verified with examples such as Table 3.1. To determine the membrane voltage when there are several types of ions in the intra- and extracellular media, to which the membrane may be permeable, an extended version of the Nernst equation must be used. This is the particular application of the Goldman-Hodgkin-Katz equation whose derivation we will now describe.
The Nernst equation evaluates the equilibrium value of the membrane voltage when the membrane is permeable to only one kind of ion or when all permeable ions have reached a Donnan equilibrium. Under physiological conditions, such an equilibrium is not achieved as can be verified with examples such as Table 3.1. To determine the membrane voltage when there are several types of ions in the intra- and extracellular media, to which the membrane may be permeable, an extended version of the Nernst equation must be used. This is the particular application of the Goldman-Hodgkin-Katz equation whose derivation we will now describe.
 For the membrane introduced above, in view of its one dimensionality, we have
For the membrane introduced above, in view of its one dimensionality, we have  ,
,  , and, using Equation 3.12, we get
, and, using Equation 3.12, we get


(3.25)


(3.26)


(3.27)


(3.28)
where ckh = concentration of the kth ion at x = h cko = concentration of the kth ion at x = 0  Both variables are defined within the membrane.
Both variables are defined within the membrane.


(3.29)





(3.30)
where b = partition coefficient ci = measurable intracellular ionic concentration co = measurable extracellular ionic concentration  The electric current density Jk can be obtained by multiplying the ionic flux jk from Equation 3.29 by Faraday's constant and valence. If, in addition, the permeability Pk is defined as
The electric current density Jk can be obtained by multiplying the ionic flux jk from Equation 3.29 by Faraday's constant and valence. If, in addition, the permeability Pk is defined as


(3.31)


(3.32)
 When considering the ion flux through the membrane at the resting state, the sum of all currents through the membrane is necessarily zero, as noted above. The main contributors to the electric current are potassium, sodium, and chloride ions. So we may write
When considering the ion flux through the membrane at the resting state, the sum of all currents through the membrane is necessarily zero, as noted above. The main contributors to the electric current are potassium, sodium, and chloride ions. So we may write


(3.33)
 By substituting Equation 3.32 into Equation 3.33, appending the appropriate indices, and noting that for potassium and sodium the valence z = +1 whereas for chloride z = -1, and canceling the constant zk˛F˛/RT, we obtain:
By substituting Equation 3.32 into Equation 3.33, appending the appropriate indices, and noting that for potassium and sodium the valence z = +1 whereas for chloride z = -1, and canceling the constant zk˛F˛/RT, we obtain:


(3.34)
 The denominator can be eliminated from Equation 3.34 by first multiplying the numerator and denominator of the last term by factor -e-FVm/RT and then multiplying term by term by 1 - e-FVm/RT. Thus we obtain
The denominator can be eliminated from Equation 3.34 by first multiplying the numerator and denominator of the last term by factor -e-FVm/RT and then multiplying term by term by 1 - e-FVm/RT. Thus we obtain


(3.35)


(3.36)


(3.37)
 By substituting 37 °C for the temperature and the Briggs logarithm (with base 10) for the Napier logarithm (to the base e), Equation 3.37 may be written as:
By substituting 37 °C for the temperature and the Briggs logarithm (with base 10) for the Napier logarithm (to the base e), Equation 3.37 may be written as:


(3.38)


(3.39)


(3.40) 
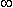 . The steady-state response is illustrated in Figure 3.9. It follows from Ohm's law that
. The steady-state response is illustrated in Figure 3.9. It follows from Ohm's law that


(3.41)


(3.42)


(3.43)


(3.44)
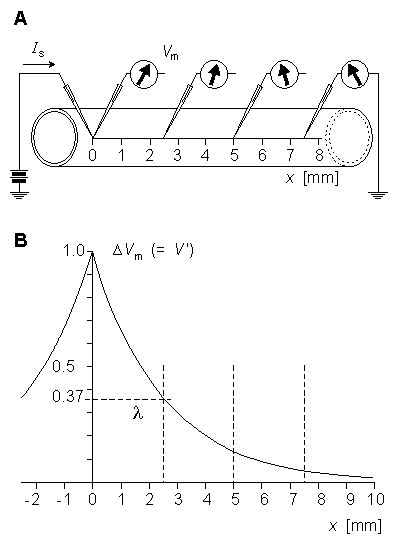
 (B) Variation of the membrane voltage as a function of distance.
(B) Variation of the membrane voltage as a function of distance.Substituting Equation 3.42 into Equation 3.44 gives:
  | (3.45) |
which is called the general cable equation.
 Under stationary and subthreshold conditions the capacitive current cmdV'/dt = 0; so that the membrane current per unit length is simply im = V'/rm; according to Ohm's law. Consequently, Equation 3.45 can be written in the form
Under stationary and subthreshold conditions the capacitive current cmdV'/dt = 0; so that the membrane current per unit length is simply im = V'/rm; according to Ohm's law. Consequently, Equation 3.45 can be written in the form
  | (3.46) |
whose solution is
  | (3.47) |
The constant l in Equation 3.47 has the dimension of length and is called the characteristic length or length constant of the axon. It is called also the space constant. The characteristic length l is related to the parameters of the axon by Equation 3.46, and is given by:
The latter form of Equation 3.48 may be written because the extracellular axial resistance ro is frequently negligible when compared to the intracellular axial resistance ri.
the constants A and B take on the values A = V'(0) and B = 0, and from Equation 3.47 we obtain the solution:
This expression indicates that V' decreases exponentially along the axon beginning at the point of stimulation (x = 0), as shown in Figure 3.9B. At x = l the amplitude has diminished to 36.8% of the value at the origin. Thus l is a measure of the distance from the site of stimulation over which a significant response is obtained. For example at x = 2l the response has diminished to 13.5%, whereas at x = 5l it is only 0.7% of the value at the origin.
3.6.3 Stimulation with a Step-Current Impulse
In this section we consider the transient (rather than steady-state) response of the axon to a subthreshold current-step input. In this case the membrane current is composed of both resistive and capacitive components reflecting the parallel RC nature of the membrane:
The left side of Equation 3.51 evaluates the total membrane current im, whereas on the right side the first term represents the resistive component (formed by the ionic currents), and the second term the capacitive current which must now be included since
which can be easily expressed as
where t = rmcm is the time constant of the membrane and l is the space constant as defined in Equation 3.48.


(3.48)  With the boundary conditions:
With the boundary conditions:




(3.49)


(3.50)
where im = the total membrane current per unit length [µA/cm axon length] imR = the resistive component of the membrane current per unit length [µA/cm axon length] imC = the capacitive component of the membrane current per unit length [µA/cm axon length]  Under transient conditions Equation 3.50 substituted into Equation 3.45 may be written:
Under transient conditions Equation 3.50 substituted into Equation 3.45 may be written:


(3.51  /
/ t
t 0 . Equation 3.51 may also be written in the form:
0 . Equation 3.51 may also be written in the form:


(3.52)


(3.53)  Here the time constant was derived for a long, thin axon corresponding to a one-dimensional problem. The time constant may be derived with a similar method also for the surface of a membrane as a two-dimensional problem. In such case instead of the variables defined "times unit length" and "per unit length", variables defined "times unit area" and "per unit area" are used. Then we obtain for the time constant t = RmCm.
Here the time constant was derived for a long, thin axon corresponding to a one-dimensional problem. The time constant may be derived with a similar method also for the surface of a membrane as a two-dimensional problem. In such case instead of the variables defined "times unit length" and "per unit length", variables defined "times unit area" and "per unit area" are used. Then we obtain for the time constant t = RmCm.
 The temporal and spatial responses of the membrane voltage for several characteristic values of x and t are illustrated in Figure 3.10. One should note that the behavior of V' as a function of x is nearly exponential for all values of t, but the response as a function of t for large values of x differs greatly from an exponential behavior (becoming S-shaped). These curves illustrate the interpretation of l, the space constant, as a measure of the spatial extent of the response to the stimulating current. For values of x/l less than around 2, t is essentially a measure of the time to reach steady state. However, for large x/l this interpretation becomes poor because the temporal curve deviates greatly from exponential. In Figure 3.10, where l = 2.5 mm, the electrode at x = 5 mm is at 2l, and the amplitude, after an interval t, has reached only 37% of steady state. Were we to examine x = 25 mm (corresponding to 5l), only 0.8% of steady-state would be reached after the interval t.
The temporal and spatial responses of the membrane voltage for several characteristic values of x and t are illustrated in Figure 3.10. One should note that the behavior of V' as a function of x is nearly exponential for all values of t, but the response as a function of t for large values of x differs greatly from an exponential behavior (becoming S-shaped). These curves illustrate the interpretation of l, the space constant, as a measure of the spatial extent of the response to the stimulating current. For values of x/l less than around 2, t is essentially a measure of the time to reach steady state. However, for large x/l this interpretation becomes poor because the temporal curve deviates greatly from exponential. In Figure 3.10, where l = 2.5 mm, the electrode at x = 5 mm is at 2l, and the amplitude, after an interval t, has reached only 37% of steady state. Were we to examine x = 25 mm (corresponding to 5l), only 0.8% of steady-state would be reached after the interval t.
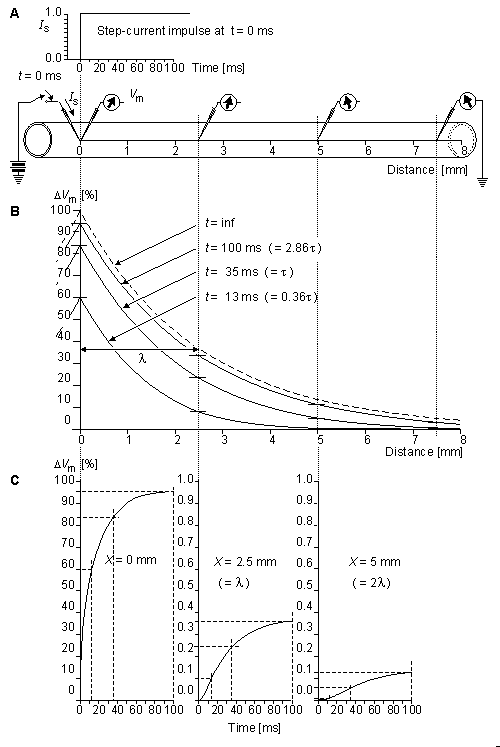
Fig. 3.10. The response of the axon to a step-current impulse.
 (A) The physical setup, including the waveform of the applied current and the placement of stimulating and recording electrodes.
(A) The physical setup, including the waveform of the applied current and the placement of stimulating and recording electrodes.
 (B) The spatial response at t = 13, 35, 100 ms; and t =
(B) The spatial response at t = 13, 35, 100 ms; and t =  . The latter curve is the steady-state response and corresponds to Equation 3.49.
. The latter curve is the steady-state response and corresponds to Equation 3.49.
 (C) The temporal response of three axial sites at x = 0, 2.5, 5 mm.
(C) The temporal response of three axial sites at x = 0, 2.5, 5 mm.
 While a closed-form solution to Equation 3.53 can be described, we have chosen to omit it from this text because of its complexity. One can find a derivation in Davis and Lorente de No (1947). Rather than include this analytical material, we have chosen instead to illustrate the temporal and spatial response of the transmembrane voltage to a current step for a range of values of l and t. This is provided in Figure 3.11.
While a closed-form solution to Equation 3.53 can be described, we have chosen to omit it from this text because of its complexity. One can find a derivation in Davis and Lorente de No (1947). Rather than include this analytical material, we have chosen instead to illustrate the temporal and spatial response of the transmembrane voltage to a current step for a range of values of l and t. This is provided in Figure 3.11.
 Specifically, Figure 3.11 describes the subthreshold transmembrane voltage response to a current step of very long duration introduced extracellularly at the center of a cable of infinite length. The response, when the current is turned on, is shown in the left-hand side of the figure, whereas the response, when the current is subsequently turned off, is on the right. The transmembrane voltage is described as a function of time for given positions of the fiber. The transmembrane voltage is also described as a function of position at given times following the application of the current or its termination. The figure is drawn from a recalculation of its quantities from the original publication of Hodgkin and Rushton (1946).
Specifically, Figure 3.11 describes the subthreshold transmembrane voltage response to a current step of very long duration introduced extracellularly at the center of a cable of infinite length. The response, when the current is turned on, is shown in the left-hand side of the figure, whereas the response, when the current is subsequently turned off, is on the right. The transmembrane voltage is described as a function of time for given positions of the fiber. The transmembrane voltage is also described as a function of position at given times following the application of the current or its termination. The figure is drawn from a recalculation of its quantities from the original publication of Hodgkin and Rushton (1946).
 Note that distance is shown normalized to the space constant , whereas time is normalized to the time constant . Normalization, such as this, results in "universal" curves that can be adapted to any actual value of and . Note also that the points on a particular voltage versus distance curve drawn at some values of t in the upper graph can also be found at the same values of t in the lower graph for the particular distance values, and vice versa. The fact that the upper and lower curves show the same phenomenon but in different dimensions is emphasized by the dotted vertical lines which indicate the corresponding location of points in the two sets of curves.
Note that distance is shown normalized to the space constant , whereas time is normalized to the time constant . Normalization, such as this, results in "universal" curves that can be adapted to any actual value of and . Note also that the points on a particular voltage versus distance curve drawn at some values of t in the upper graph can also be found at the same values of t in the lower graph for the particular distance values, and vice versa. The fact that the upper and lower curves show the same phenomenon but in different dimensions is emphasized by the dotted vertical lines which indicate the corresponding location of points in the two sets of curves.
 Table 3.2 lists measured values of characteristic lengths and time constants for several axons for several different species. A significant variation from species to species is seen.
Table 3.2 lists measured values of characteristic lengths and time constants for several axons for several different species. A significant variation from species to species is seen.
| |||||||||||||||||||||||||||||||||
 resistance and capacitance per unit length by use of the following:
resistance and capacitance per unit length by use of the following:
 Rm = 2parm Rm = 2parm | (3.54) |
 Cm = cm/(2pa) Cm = cm/(2pa) | (3.55) |
| where | Rm | = specific resistance of the membrane (membrane resistance times unit area) [kW·cm2] |
| rm | = membrane resistance times unit length [kW·cm axon length] | |
| Cm | = specific capacitance of the membrane (membrane capacitance per unit area) [µF/cm˛] | |
| cm | = membrane capacitance per unit length [µF/cm axon length] | |
| a | = fiber radius [cm]. |
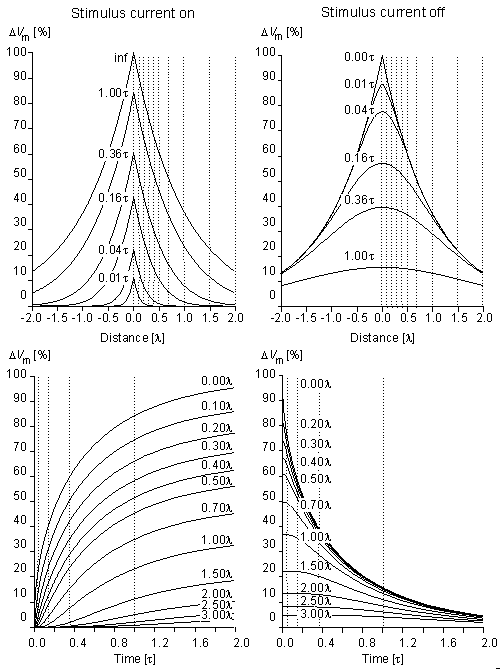
Fig. 3.11. Subthreshold transmembrane voltage response to a step current of very long duration at different instants of time (upper graphs) and at different distances from the sites of stimulation (lower graphs). The responses when the current is turned on and off are shown in the left and right sides of the figure, respectively.
  | (3.56) |
| where | V' | = change in the membrane voltage [mV] |
| Is | = stimulus current per unit area [µA/cm˛] | |
| Rm | = membrane resistance times unit area [kW·cm˛] | |
| t | = stimulus time [ms] | |
| t | = membrane time constant = RmCm [ms] | |
| Cm | = membrane capacitance per unit surface [µF/cm˛] |
 Unfortunately, this simple analysis cannot be applied to cells with other shapes (e.g., the fiberlike shape of excitable cells), where the response to a stimulating current follows that governed by Equation 3.53 and described in Figure 3.11. However, Equation 3.56 could still be viewed as a first-order approximation based on a lumped-parameter representation of what is actually a distributed-parameter structure. Following this argument, in Figure 3.12 we have assumed that a long fiber can be approximated by just a single (lumped) section, hence leading to an equation of the type described in Equation 3.56. A characteristic response based on Equation 3.56 is also shown in Figure 3.12..
Unfortunately, this simple analysis cannot be applied to cells with other shapes (e.g., the fiberlike shape of excitable cells), where the response to a stimulating current follows that governed by Equation 3.53 and described in Figure 3.11. However, Equation 3.56 could still be viewed as a first-order approximation based on a lumped-parameter representation of what is actually a distributed-parameter structure. Following this argument, in Figure 3.12 we have assumed that a long fiber can be approximated by just a single (lumped) section, hence leading to an equation of the type described in Equation 3.56. A characteristic response based on Equation 3.56 is also shown in Figure 3.12..
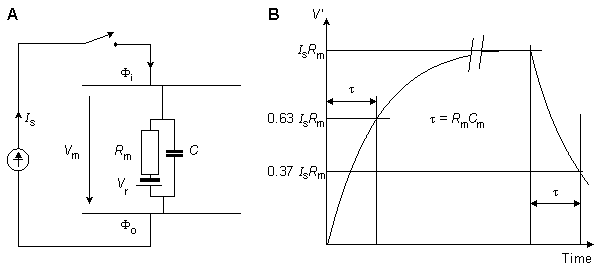
Fig. 3.12. The derivation of the strength-duration curve.
 (A) An approximate lumped-parameter RC-network which replaces the actual distributed parameter structure.
(A) An approximate lumped-parameter RC-network which replaces the actual distributed parameter structure.
 (B) The response of the network to a current pulse of magnitude Is is exponential and is shown for a pulse of very long duration.
(B) The response of the network to a current pulse of magnitude Is is exponential and is shown for a pulse of very long duration.
The membrane is assumed to be activated if its voltage reaches the threshold value. We consider this condition if we substitute V' = DVth into Equation 3.56, where Vth is the change in the resting voltage needed just to reach the threshold voltage. Equation 3.56 may now be written in the form:


(3.57)  The smallest current that is required for the transmembrane voltage to reach threshold is called the rheobasic current. With this stimulus current, the required stimulus duration is infinite. Because the rheobasic current is given by Irh = DVth/Rm, the strength-duration curve takes on the form:
The smallest current that is required for the transmembrane voltage to reach threshold is called the rheobasic current. With this stimulus current, the required stimulus duration is infinite. Because the rheobasic current is given by Irh = DVth/Rm, the strength-duration curve takes on the form:


(3.58)  The strength-duration curve is illustrated in Figure 3.13. Here the stimulus current is normalized so that the rheobasic current has the strength of unity. (Note again, that this result is derived for a space-clamp situation.)
The strength-duration curve is illustrated in Figure 3.13. Here the stimulus current is normalized so that the rheobasic current has the strength of unity. (Note again, that this result is derived for a space-clamp situation.)
 The time needed to reach the threshold voltage with twice the rheobasic stimulus current is called chronaxy. For the relation between chronaxy and the membrane time constant, Equation 3.57 can be written as:
The time needed to reach the threshold voltage with twice the rheobasic stimulus current is called chronaxy. For the relation between chronaxy and the membrane time constant, Equation 3.57 can be written as:


(3.59)


(3.60)


(3.61)  If the stimulus current is twice rheobasic current, then Is = 2(DVth/Rm), and we obtain for chronaxy:
If the stimulus current is twice rheobasic current, then Is = 2(DVth/Rm), and we obtain for chronaxy:


(3.62)
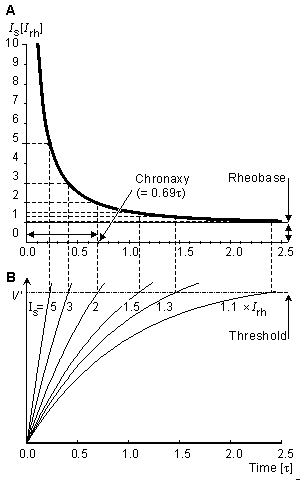
Fig. 3.13. (A) Strength-duration curve. The units are relative.
 (B) The subthreshold transient response prior to excitation.
(B) The subthreshold transient response prior to excitation.
The analytical results above are approximate for several reasons. First, the excitable tissue cannot normally be well approximated by a lumped R since such elements are actually distributed. (In a space-clamp stimulation the membrane can be more accurately represented with a lumped model.) Also the use of a linear model is satisfactory up to perhaps 80% of the threshold, but beyond this the membrane behaves nonlinearly. Another approximation is the idea of a fixed threshold; in a subsequent chapter, we describe accommodation, which implies a threshold rising with time.
 In a particular situation, a strength-duration curve can be found experimentally. In this case, rheobase and chronaxy are more realistic measures of the stimulus-response behavior. This type of data for chronaxy is given in Table 3.3, which lists chronaxies measured for various nerve and muscle tissues. Note that, in general, the faster the expected response from the physiological system, the shorter the chronaxy value.
In a particular situation, a strength-duration curve can be found experimentally. In this case, rheobase and chronaxy are more realistic measures of the stimulus-response behavior. This type of data for chronaxy is given in Table 3.3, which lists chronaxies measured for various nerve and muscle tissues. Note that, in general, the faster the expected response from the physiological system, the shorter the chronaxy value.
|